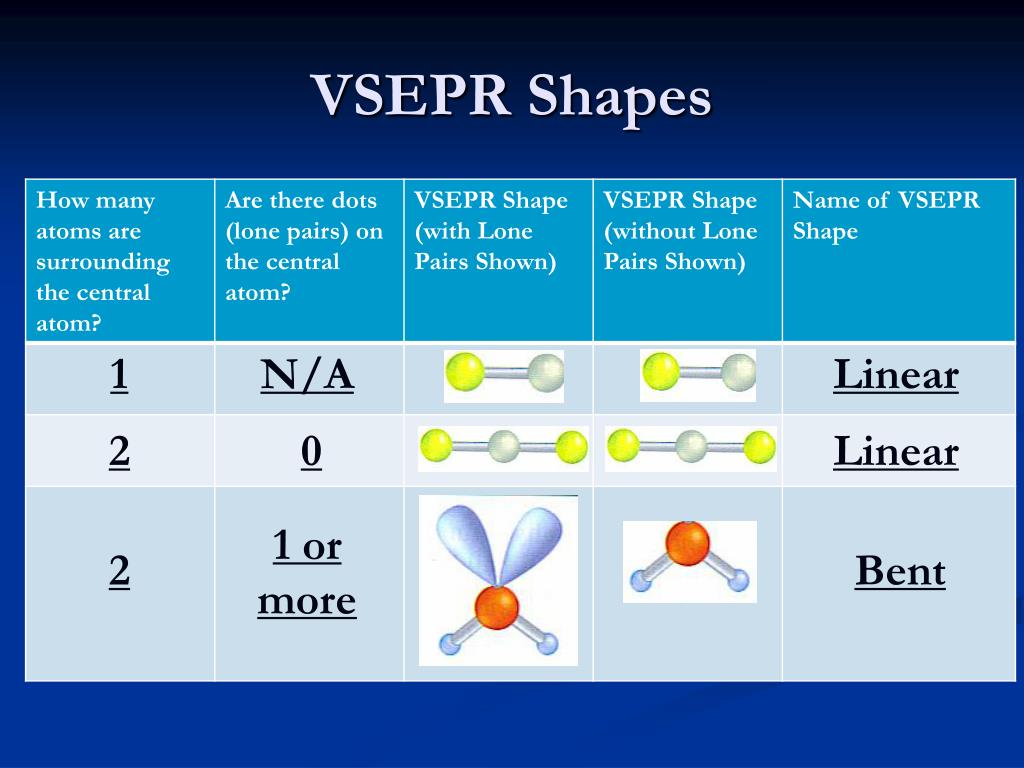
The electron-pair geometries will be the same as the molecular structures when there are no lone electron pairs around the central atom, but they will be different when there are lone pairs present on the central atom. VSEPR and Molecular Shapes : Valence Shell Electron Pair Repulsion (VSEPR) Theory is a simple, qualitative model that allows the prediction of an approximate molecular shape, given a valid Lewis structure of a molecule. The structure that includes only the placement of the atoms in the molecule is called the molecular structure. 
VSEPR Theory states: i) electron pairs repel to. We differentiate between these two situations by naming the geometry that includes all electron pairs the electron-pair geometry. It is the mutual repulsion between these electron pairs which gives the molecule its shape and bond angles. Molecular structure describes the location of the atoms, not the electrons. The electron-pair geometries describe all regions where electrons are located, bonds as well as lone pairs. It is important to note that electron-pair geometry around a central atom is not the same thing as its molecular structure. Using this as an example, the drug a molecule with a specific shape will commonly interact with a larger molecule, such as a protein. One important area is how drugs work in the body. The electrostatic repulsion of these electrons is reduced when the various regions of high electron density assume positions as far from each other as possible. The shape of a molecule affects how it interacts with other molecules and that in turn can give rise to all kinds of interesting phenomena. Although the VSEPR model is a simple and useful method for qualitatively predicting the structures of a wide range of compounds, it is not infallible. The electrons in the valence shell of a central atom form either bonding pairs of electrons, located primarily between bonded atoms, or lone pairs. The VSEPR model assumes that electron pairs in the valence shell of a central atom will adopt an arrangement that minimizes repulsions between these electron pairs by maximizing the distance between them. In the water molecule, two of the electron pairs are lone pairs rather than bonding pairs. As for methane and ammonia, the domain geometry for a molecule with four electron pairs is tetrahedral. Valence shell electron-pair repulsion theory (VSEPR theory) enables us to predict the molecular structure, including approximate bond angles around a central atom, of a molecule from an examination of the number of bonds and lone electron pairs in its Lewis structure. The VSEPR theory predicts the correct shape based on three groups of bonding electrons and no lone pairs on electrons on the central carbon atom. The Valence Shell Electron Pair Repulsion (VSEPR) Theory can be used to predict the shapes of molecules based on the number of electron pairs around the. A water molecule consists of two bonding pairs and two lone pairs (see Figure 10 ).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
